
Recent Progress in the Use of Pd-Catalyzed C-C Cross-Coupling Reactions in the Synthesis of Pharmaceutical Compounds

Molecular structure of C2. Selected bond distances (Å): Pd(1)−C(11)... | Download Scientific Diagram

Are Imidazoles Versatile or Promiscuous in Reactions with Organophosphates? Insights from the Case of Parathion

Exploring Green Solvents Associated to Pd/C as Heterogeneous Catalyst for Direct Arylation of Heteroaromatics with Aryl Bromides - Mao - 2018 - Advanced Synthesis & Catalysis - Wiley Online Library

Palladium‐ and Copper‐Mediated Direct C‐2 Arylation of Azoles — Including Free (NH)‐Imidazole, ‐Benzimidazole and ‐Indole — Under Base‐Free and Ligandless Conditions - Bellina - 2006 - European Journal of Organic Chemistry - Wiley Online Library
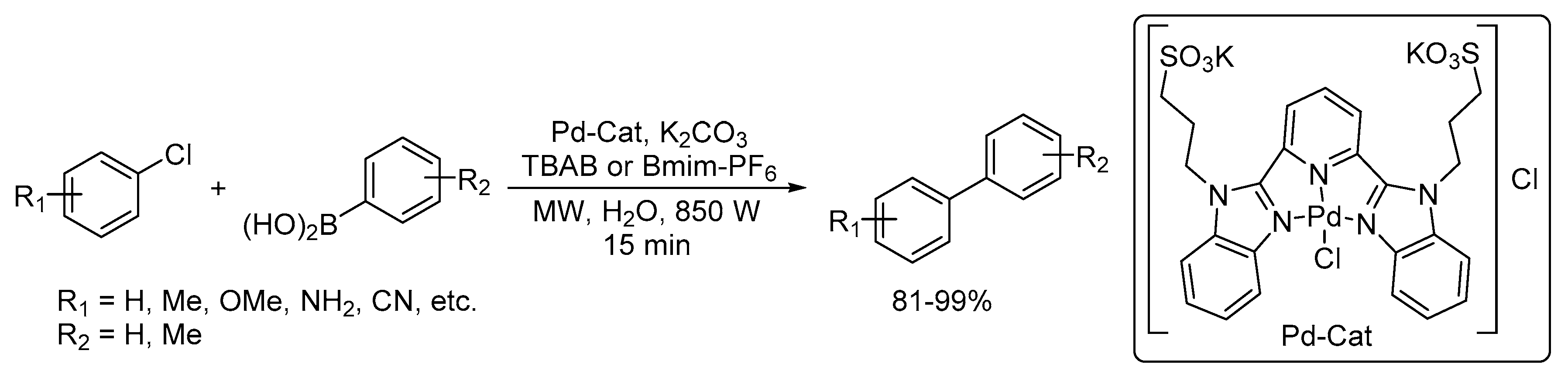
Catalysts | Free Full-Text | Microwave-Assisted Palladium-Catalyzed Cross-Coupling Reactions: Generation of Carbon–Carbon Bond | HTML
![Molecules | Free Full-Text | Pharmacological Potential and Synthetic Approaches of Imidazo[4,5-b]pyridine and Imidazo[4,5-c]pyridine Derivatives | HTML Molecules | Free Full-Text | Pharmacological Potential and Synthetic Approaches of Imidazo[4,5-b]pyridine and Imidazo[4,5-c]pyridine Derivatives | HTML](https://www.mdpi.com/molecules/molecules-22-00399/article_deploy/html/images/molecules-22-00399-sch013.png)
Molecules | Free Full-Text | Pharmacological Potential and Synthetic Approaches of Imidazo[4,5-b]pyridine and Imidazo[4,5-c]pyridine Derivatives | HTML

Table 1 from Interaction Between Pd(RaaiR/)Cl2 and HQ: Reaction Dynamics and Mechanism (RaaiR/ = 1-alkyl-2-(arylazo)imidazole; HQ = 8-Quinolinol) | Semantic Scholar

Regiocontrolled Synthesis of 1,2‐Diaryl‐1H‐imidazoles by Palladium‐ and Copper‐Mediated Direct Coupling of 1‐Aryl‐1H‐imidazoles with Aryl Halides under Ligandless Conditions - Bellina - 2006 - European Journal of Organic Chemistry - Wiley Online Library

Cycloheptyl substituted N-heterocyclic carbene PEPPSI-type palladium complexes with different N-coordinated ligands: Involvement in Suzuki-Miyaura reaction - ScienceDirect
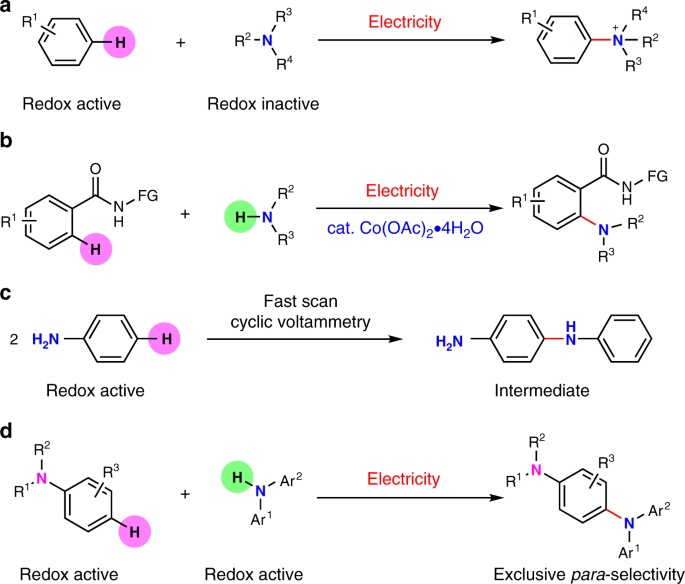
Electrooxidative para -selective C–H/N–H cross-coupling with hydrogen evolution to synthesize triarylamine derivatives | Nature Communications

US20110028733A1 - Process for the preparation of 5-(2-ethyl-dihydro-1h-inden-2-yl)-1h-imidazole and salts thereof - Google Patents

PDF) Efficient hydroarylation of terminal alkynes with sodium tetraphenylborate performed in water under mild conditions
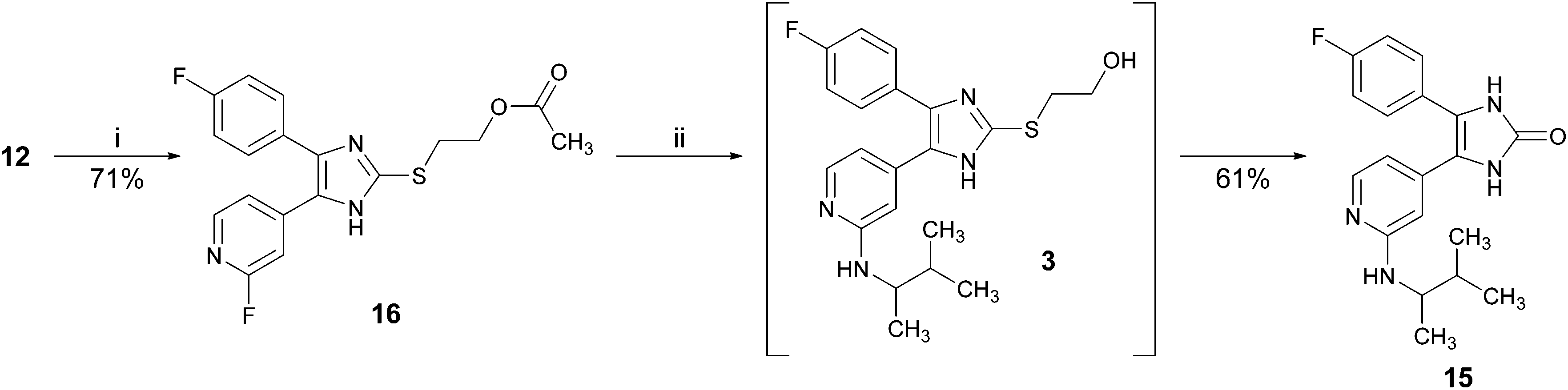
An optimized and versatile synthesis to pyridinylimidazole-type p38α mitogen activated protein kinase inhibitors - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C5OB01505G
![Unexpected photochemical transformation of imidazole derivatives containing the 5-hydroxy-2-methyl-4H-pyran-4-one moiety. Environmentally friendly method for the synthesis of substituted imidazo[1,5-a]pyridine-5,8-diones - Tetrahedron Lett. - X-MOL Unexpected photochemical transformation of imidazole derivatives containing the 5-hydroxy-2-methyl-4H-pyran-4-one moiety. Environmentally friendly method for the synthesis of substituted imidazo[1,5-a]pyridine-5,8-diones - Tetrahedron Lett. - X-MOL](https://xpic.x-mol.com/20190826%2F10.1016_j.tetlet.2019.151080.jpg)
Unexpected photochemical transformation of imidazole derivatives containing the 5-hydroxy-2-methyl-4H-pyran-4-one moiety. Environmentally friendly method for the synthesis of substituted imidazo[1,5-a]pyridine-5,8-diones - Tetrahedron Lett. - X-MOL

Imidazole-aryl coupling reaction via CH bond activation catalyzed by palladium supported on modified magnetic reduced graphene oxide in alkaline deep eutectic solvent - ScienceDirect
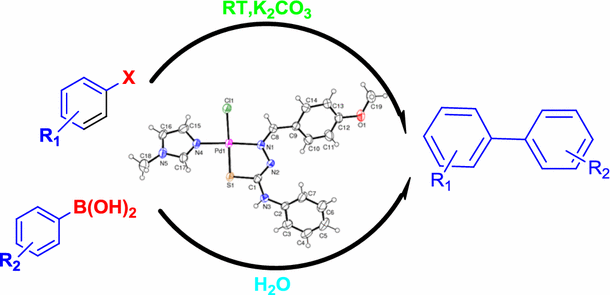
A thiosemicarbazone–palladium(II)–imidazole complex as an efficient pre-catalyst for Suzuki–Miyaura cross-coupling reactions at room temperature in aqueous media | SpringerLink
![Synthesis, Structural Characterization, and Coordination Chemistry of (Trineopentylphosphine)palladium(aryl)bromide Dimer Complexes ([(Np3P)Pd(Ar)Br]2) - Inorg. Chem. - X-MOL Synthesis, Structural Characterization, and Coordination Chemistry of (Trineopentylphosphine)palladium(aryl)bromide Dimer Complexes ([(Np3P)Pd(Ar)Br]2) - Inorg. Chem. - X-MOL](https://xpic.x-mol.com/20190926%2F10.1021_acs.inorgchem.9b02164.jpg)
Synthesis, Structural Characterization, and Coordination Chemistry of (Trineopentylphosphine)palladium(aryl)bromide Dimer Complexes ([(Np3P)Pd(Ar)Br]2) - Inorg. Chem. - X-MOL
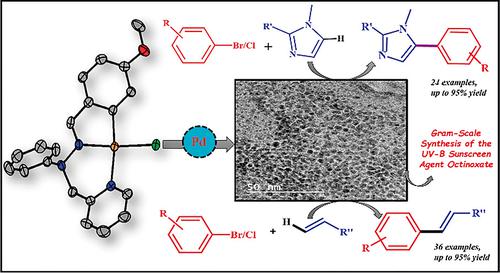
Palladium‐Based Catalysts Supported by Unsymmetrical XYC–1 Type Pincer Ligands: C5 Arylation of Imidazoles and Synthesis of Octinoxate Utilizing the Mizoroki–Heck Reaction - Eur. J. Inorg. Chem. - X-MOL

Dimetallic Palladium‐NHC Complexes: Synthesis, Characterization, and Catalytic Application for Direct C−H Arylation Reaction of Heteroaromatics with Aryl Chlorides - Lee - 2020 - Advanced Synthesis & Catalysis - Wiley Online Library
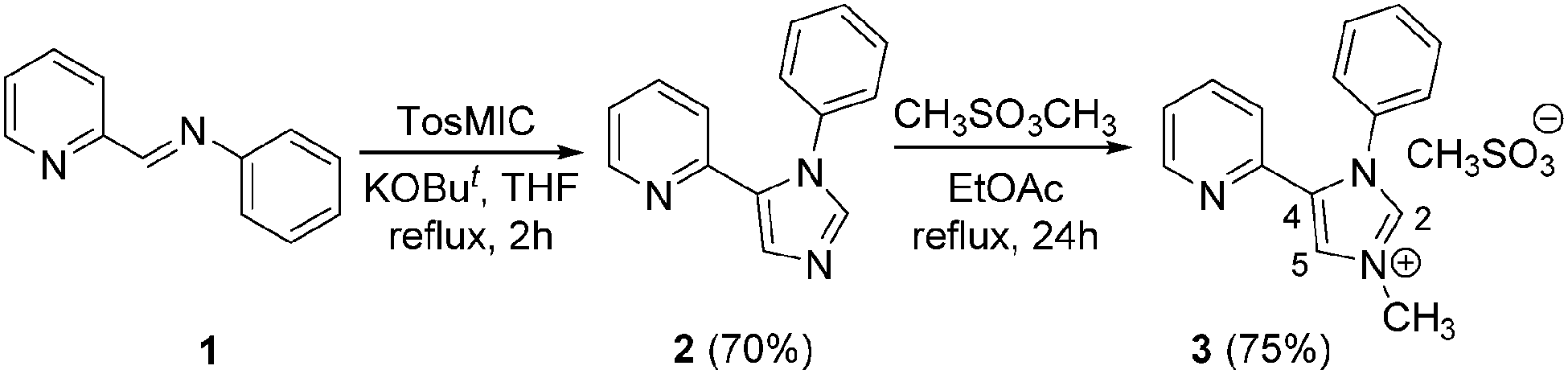
Chelation assistance as a tool for the selective preparation of an imidazole-based mesoionic palladium carbene complex - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC07732F

Tetranuclear Palladium Complexes of Abnormal N‐Heterocyclic Carbene Ligands and their Catalytic Activities in Mizoroki‐Heck Coupling Reaction of Electron‐Rich Aryl Chlorides - Lee - 2019 - Advanced Synthesis & Catalysis - Wiley Online Library

